When you pick up a prescription for a generic drug like metformin or lisinopril, you probably don’t think about who made it - or how many other companies are making the same thing. But that’s exactly what keeps your co-pay low. The more manufacturers that produce the same generic drug, the lower the price drops. It’s simple economics: more players in the market mean less room for anyone to charge high prices.
Why competition matters in generic drugs
Generic drugs are chemically identical to brand-name versions. They work the same way, have the same side effects, and are approved by the FDA as safe and effective. But while brand-name drugs have patent protection, generics enter the market after those patents expire. And when they do, prices don’t just drop - they plummet.A 2021 study published in JAMA Network Open tracked 50 brand-name drugs and their generic versions. The results were clear: the first generic competitor brought down prices by an average of 17%. With two manufacturers, prices fell 39.5%. With three, they dropped 52.5%. And when four or more companies started making the same drug, prices crashed by 70.2% compared to the original brand price.
That’s not theory. That’s real savings. In 2022 alone, the FDA estimated that 742 new generic approvals saved the U.S. healthcare system $14.5 billion in one year. Over the past decade, generics saved the system nearly $1.7 trillion. That’s money that goes back into patients’ pockets, insurance premiums, and public health programs.
How many manufacturers actually exist?
Not all generic drugs have equal competition. Some have dozens of makers. Others have just one or two.Oral pills - like cholesterol meds, blood pressure drugs, or antibiotics - are the most competitive. For example, metformin, a diabetes drug, is made by at least eight different companies. That’s why you can buy a 90-day supply for under $10 at many pharmacies. But look at a drug like levetiracetam, an epilepsy medication. In 2023, the number of manufacturers dropped from five to two. Prices jumped 300% overnight. Patients had to switch drugs or pay hundreds more per month.
According to a 2017 study from the National Bureau of Economic Research, over half of all generic drugs in the U.S. have at most two manufacturers. And 40% have just one. That’s a problem. When only one company makes a drug, prices stay high. When two companies make it, prices drop only 10-15%. But once you hit three or more, the real savings kick in.
Where competition fails: injectables, biologics, and shortages
Not all drugs follow the same rules. Injected or infused generics - like those used in hospitals - see far less competition. Why? Manufacturing them is harder and more expensive. Fewer companies are willing to enter that market.Biosimilars - the generic version of complex biologic drugs like Humira or Enbrel - have struggled to gain traction. Even when approved, they often don’t drive prices down like traditional generics. A JAMA study found that if biosimilars were treated like regular generics under Medicare, spending on those drugs could have dropped 27% between 2015 and 2019. But they’re not. Regulatory hurdles, patent games, and payer resistance keep them from competing fairly.
And then there’s the shortage crisis. When a drug has only one or two manufacturers and one of them shuts down production - due to quality issues, financial strain, or regulatory problems - the entire supply chain collapses. Patients suddenly can’t get their meds. Prices spike. In 2022, a single manufacturer’s exit caused a 500% price jump for a common generic antibiotic. That’s not an anomaly. It’s becoming common.
Who’s behind the scenes? PBMs, wholesalers, and consolidation
You might think more manufacturers = lower prices. But the system is more complicated.Pharmacy Benefit Managers (PBMs) - middlemen that negotiate drug prices for insurers - have consolidated power. They control which generics get placed on formularies. Sometimes, they favor one manufacturer over others, even if the price isn’t the lowest. This distorts competition.
Wholesalers and distributors also add layers of markup. A 2019 FDA analysis found that while manufacturer prices dropped sharply with competition, wholesale invoice prices didn’t fall as much. That’s because the middlemen take bigger cuts. The savings aren’t always passed on to you.
Meanwhile, small generic manufacturers are being bought up. Between 2014 and 2016, nearly 100 mergers happened in the generic drug space. These aren’t flashy deals like Big Pharma acquisitions. They’re quiet, low-profile buys - often of companies making just one or two drugs. The result? A market that looks competitive on paper, but in reality, a handful of corporations control most of the supply.
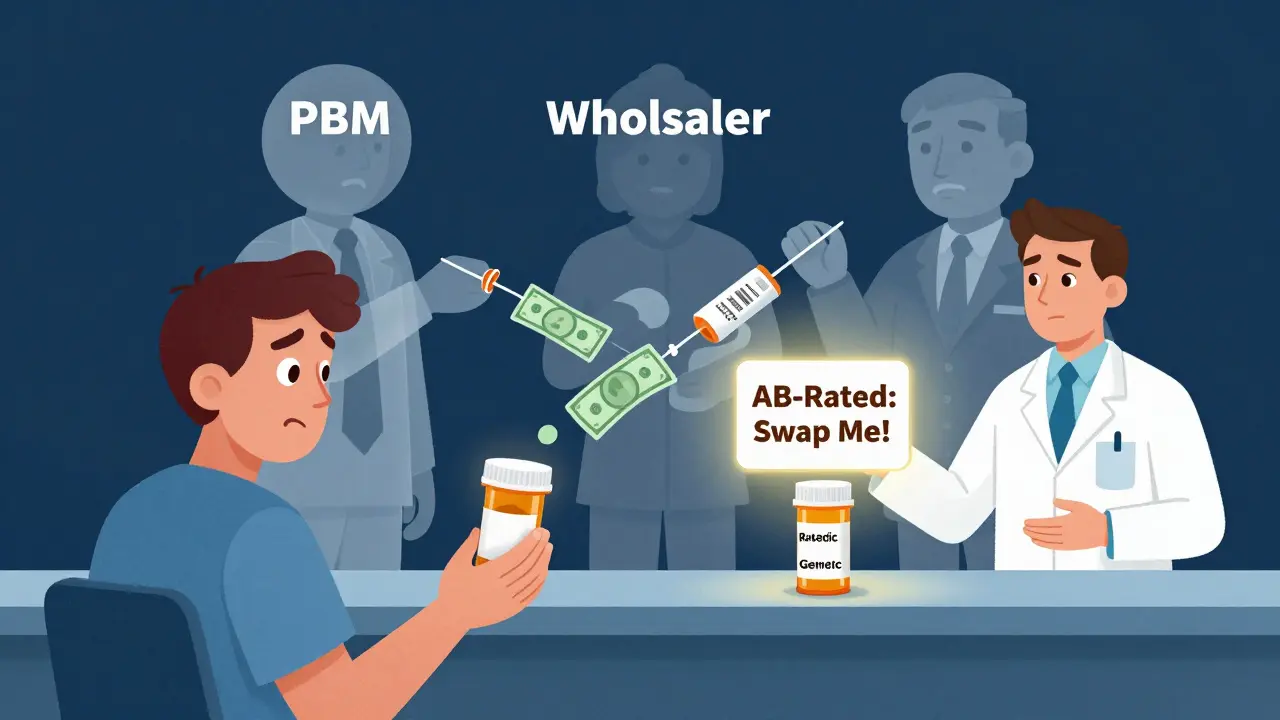
What you can do to save money
You don’t need to be an economist to use this system to your advantage.- Ask your pharmacist: “Is there another generic maker for this drug?” If your prescription says “generic,” it might be made by only one company. Switching to another brand of the same generic can cut your cost.
- Use GoodRx or similar tools. They compare prices across pharmacies and often show which generic version is cheapest.
- Check the FDA’s Orange Book. It lists therapeutic equivalence ratings. Drugs marked “AB” are interchangeable. If your drug is AB-rated, your pharmacist can legally swap it for another brand - even if your doctor didn’t specify.
- Ask your doctor about therapeutic substitution. In many states, pharmacists can switch your generic without asking - but only if the drugs are rated equivalent.
For example, if you’re taking a generic version of losartan made by Company A, and Company B makes the same drug for $5 less per month, your pharmacist can switch you - unless your doctor specifically says “do not substitute.”
The future: Will competition keep working?
The system works - when it’s allowed to. But it’s under pressure.The FDA’s Drug Competition Action Plan, launched in 2017, tried to speed up approvals and block anti-competitive tactics. The CREATES Act of 2019 aimed to stop brand manufacturers from delaying generic entry by refusing to share samples or blocking distribution. These are good steps.
But consolidation continues. The NBER researchers warn that many mergers in the generic space fly under the radar. The FTC and DOJ rarely challenge them because the companies are small. But small companies, when combined, can control entire markets.
Without intervention, we risk a future where lifesaving generics - like insulin, epinephrine, or antibiotics - are made by only one or two companies. And when that happens, prices rise. Supply chains break. Patients suffer.
The lesson is clear: competition isn’t just good policy - it’s essential medicine. More manufacturers mean lower prices. Fewer manufacturers mean higher costs and real danger. The next time you pay $12 for a generic pill, remember: it’s not luck. It’s competition.
Why do generic drug prices drop so much when more companies enter the market?
When multiple manufacturers make the same generic drug, they compete on price. The first competitor typically lowers prices by about 17%. With two, it’s around 40%. With three or more, prices fall by over 50%. When four or more companies are involved, prices often drop by 70% or more compared to the original brand-name drug. This happens because each manufacturer tries to win market share by offering lower prices, and the pressure keeps driving prices down.
Can a generic drug have only one manufacturer?
Yes. In fact, about 40% of generic drugs in the U.S. are made by only one company. When that happens, there’s no competition to push prices down. The single manufacturer can charge higher prices without fear of losing customers. This is especially common with older, low-margin drugs or those that are harder to produce, like injectables or complex formulations. If that one company shuts down production or faces a shortage, prices can spike by 300-500% overnight.
Why aren’t biosimilars driving prices down like regular generics?
Biosimilars are complex drugs made from living cells, unlike traditional generics that are simple chemical copies. Manufacturing them is expensive, and regulatory approval is slower. Many brand-name biologic manufacturers use legal tactics - like patent extensions or exclusive contracts with insurers - to delay biosimilar entry. Even when biosimilars are approved, they often don’t get placed on pharmacy formularies, so patients can’t access them. As a result, biosimilars have had far less impact on lowering prices than traditional generics.
How can I find out which company makes my generic drug?
Check the label on your prescription bottle - it should list the manufacturer’s name. You can also ask your pharmacist. Some pharmacies show this information on receipts or in online portals. If you want to compare prices across manufacturers, use tools like GoodRx, which often show different prices for the same generic depending on the maker. You can also look up the drug in the FDA’s Orange Book, which lists approved manufacturers and their therapeutic equivalence ratings.
Does my insurance plan affect which generic I get?
Yes. Your insurance plan’s formulary - the list of approved drugs - determines which generics are covered and at what cost. Some plans prefer one manufacturer over another, even if the price difference is small. This is often due to deals between the plan and a specific drugmaker or pharmacy benefit manager (PBM). If your plan doesn’t cover a cheaper generic version, you may have to pay more. You can ask your pharmacist to check if a different generic version is covered at a lower cost, or request a formulary exception from your insurer.









11 Comments
Generic drugs are wild when you think about it. One pill, same chemistry, same effect, but price varies like crazy based on who made it. I had to switch my blood pressure med last year because my pharmacy switched manufacturers without telling me. Cost dropped from $45 to $12. No one warned me. Just happened. That’s the system.
Competition isn’t just economic theory-it’s survival. When you have eight companies making metformin, they’re not just competing on price, they’re competing on reliability, on delivery timelines, on quality control. The moment one drops out, the others don’t fill the gap-they raise prices. It’s not greed, it’s structural vulnerability. We treat medicine like a commodity, but it’s not. It’s life support.
And yet, we let consolidation happen quietly. A small generic maker gets bought by a PBM-owned shell corporation, and suddenly, there’s only one ‘brand’ left, even though it’s still labeled ‘generic.’ The FDA approves it. The pharmacy dispenses it. The patient pays more. No one questions it because the label hasn’t changed.
This isn’t capitalism. It’s a rigged simulation of capitalism. We need transparency. Not just in pricing, but in ownership. Who owns what? Who controls the supply chain? The Orange Book lists manufacturers, but not the corporate parents behind them. That’s the real gap.
And don’t get me started on injectables. Those aren’t just hard to make-they’re politically ignored. Hospitals are forced into monopsonies. One supplier. One price. No alternatives. Patients die waiting for a vial that doesn’t exist. That’s not a market failure. That’s a policy failure.
We need to treat drug manufacturing like infrastructure. Not a profit center. Not a side hustle for contract labs. Infrastructure. With oversight. With redundancy. With public accountability.
Stop pretending this is about patients. It’s about PBMs and big insurers gaming the system. They don’t want competition-they want control. The cheapest generic doesn’t always get on the formulary. The one with the biggest kickback does. You think you’re saving money? You’re just paying the middleman under a different name.
And don’t even get me started on how Congress lets this happen. Every time someone tries to pass a bill to force transparency, Big Pharma buys off the committee. You think the FDA’s Drug Competition Action Plan actually changed anything? It’s a PR stunt. The same 5 corporations control 80% of generics now. They just changed their logos.
They’re lying about the 70% price drop. It’s all smoke and mirrors. The real savings? Half of it gets swallowed by insurance middlemen. The rest? Goes to the manufacturers who then raise prices on the next drug because they know they can. This whole system is designed to make you think you’re getting a deal while they quietly consolidate power. And the FDA? They’re asleep. Or worse-complicit.
Who owns the companies making your pills? Look it up. You’ll find most are subsidiaries of the same 3 Chinese conglomerates. And we let that happen because we’re too busy yelling about tariffs to ask who controls our medicine.
One day, the supply chain breaks. Not because of a factory shutdown. Because someone flips a switch in Shanghai. And then you’ll see what real scarcity looks like.
Let’s cut through the noise. The 70% price drop only applies to oral solids. Injectables? Biologics? You’re talking 10-15% savings max. And biosimilars? They’re not even in the same league. The FDA’s Orange Book is a joke. AB-rated doesn’t mean identical. It means ‘close enough.’ And for some patients, that difference is life or death. You think switching from one metformin brand to another is safe? Try it when you’re diabetic. Your A1C doesn’t care about regulatory equivalence.
The real problem? We treat pharmaceuticals like consumer goods. They’re not. They’re biological interventions. The body doesn’t care if the pill is made by Company A or B. But your kidneys? Your liver? They notice. And no one tracks long-term outcomes.
Y’all need to chill. I’m from Nigeria, and we don’t even have access to generics half the time. Here in the US you’re arguing over $5 differences on pills that cost $12. In Lagos, we’re waiting 6 months for insulin. If you had to choose between buying your meds or feeding your kids, you’d be grateful for ANY generic, no matter the maker.
Stop overthinking. The fact that you can get metformin for $10 is a miracle. Celebrate that. Then fight for the next person who can’t.
Let me add something from India. We produce over 40% of the world’s generic drugs. And yes, we’re not perfect. Quality varies. But the competition here? It’s brutal. Dozens of factories making the same drug. Prices are insane. A 30-day supply of lisinopril? $1.50. Not because of regulation. Because there are 17 companies fighting over market share.
Here’s the thing nobody talks about: Indian manufacturers don’t have PBMs. No middlemen. No formulary games. They sell directly to distributors. That’s why the price drops are so steep. The US system is over-engineered. We added layers. Layers that siphon off savings.
Maybe we need to stop pretending American pharmacy chains are the solution. Maybe we need to look at how the rest of the world does it.
I’ve been on a generic antidepressant for 8 years. I’ve switched manufacturers five times. Sometimes the pill looks different. Sometimes the side effects change. Not dramatically. But enough to notice. The body remembers. And doctors? They rarely ask. It’s like we’re just supposed to accept it.
I started tracking my mood logs after the third switch. I realized: the version made by Teva felt ‘heavier.’ The one from Mylan felt ‘sharper.’ I couldn’t explain it. But my sleep and anxiety levels changed. I asked my pharmacist. She said, ‘They’re bioequivalent.’
But what does that really mean? If two drugs are 90% identical, and you’re someone whose brain is already fragile… is that enough?
I don’t have answers. But I think we need to stop pretending biology is a spreadsheet.
You all are missing the point. The real issue is the FDA’s approval backlog. It takes 3-5 years to get a generic approved. Meanwhile, brand manufacturers use every loophole in the book to delay entry. The CREATES Act was a step, but it’s toothless. Why? Because enforcement is weak. The FTC doesn’t have the resources. And Congress won’t fund it. So we get 40% of generics with one manufacturer. Because it’s easier to let one company monopolize than to fix the system.
And yes, consolidation is happening. But no one wants to admit it’s because small manufacturers can’t survive without being bought out. They can’t compete with the scale of big players. And without scale, they can’t afford compliance. It’s a death spiral.
Fix the approval process. Fund the FDA. Stop pretending this is about free markets. It’s about bureaucratic failure.
It’s inspiring to see how much good can come from competition. The fact that millions of Americans can afford their prescriptions today because multiple manufacturers entered the market is a triumph of policy and market forces working together. We should be proud of this system, even with its flaws.
Let’s not let fear or cynicism blind us to the real progress: $1.7 trillion saved over a decade. That’s not just numbers-that’s lives. Families. Careers. Dreams.
Yes, there are gaps. Yes, consolidation is a concern. But the answer isn’t to dismantle the system. It’s to strengthen it-with transparency, oversight, and smart policy. We’ve done it before. We can do it again.
Did you know the FDA approves generics based on bioequivalence studies done in healthy volunteers? Not patients. Not people with liver disease. Not diabetics. Not pregnant women. So your ‘identical’ generic was tested on a 25-year-old guy who smokes and drinks energy drinks. And now you’re taking it? Good luck.
And who funds those studies? The manufacturer. So they pick the healthiest volunteers. The cleanest lab conditions. The most favorable metrics. It’s not science. It’s theater.
And don’t get me started on the Chinese supply chain. Most active pharmaceutical ingredients come from there. One factory shutdown. One inspection failure. And your entire class of drugs vanishes. No backup. No redundancy. Just… gone.
They’re not selling you medicine. They’re selling you a gamble.